What to Expect with 340B in 2019 – The Debate’s Not Over

At AlliantRx, we know drug pricing remains a top priority for you, your pharmacy’s business, and your patients. We’re committed to keeping you in the loop of key issues that can affect drug pricing and help you navigate them accordingly.
Over the past year, Medicare and Medicaid’s 340B drug pricing program has become a hot-button issue for Congressional policymakers and various healthcare stakeholders. In the eyes of many, this program is a key tool in the ongoing, multi-disciplinary efforts to lower U.S. drug pricing.
While hospitals are most frequently talked about in 340B affairs, benefits and issues can affect the volume of a pharmacy’s contracts with said 340B hospitals, as the law designates that any retail pharmacy can serve as a contracted pharmacy.
In early 2018, significant cuts were made to the program, imposing a reduction in reimbursement rates of nearly 30%. Then in late 2018, a federal court ruled against these cuts, stating that CMS acted outside of its authority.
As of February 2019, the court hasn’t given orders on how it will effectuate the ruling. It’s also still unclear whether CMS will appeal the decision, though many industry experts expect an appeal.
The state of the issue leaves us with a big question: What’s in store for 340B?
The Rise and Boom of 340B
 In 1992, the 340B program was introduced to incentivize healthcare organizations to better treat uninsured and low-income patients in some of the nation’s most underserved areas.
In 1992, the 340B program was introduced to incentivize healthcare organizations to better treat uninsured and low-income patients in some of the nation’s most underserved areas.
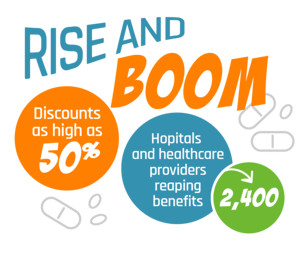
The program provides these organizations with discounts as high as 50% for certain outpatient drugs. The savings are then meant to be reinvested into providing better care to these vulnerable populations by improving services like treatment for chronic conditions, immunizations, trauma care, care for children and expecting mothers, and more.
Program participation has skyrocketed in recent years. The U.S. Government Accountability Office reports a 60% increase in participating hospitals from 2011 to 2016, with more than 2,400 hospitals and other healthcare providers reaping benefits.
Left Hanging in the Balance
 In early 2018, CMS reduced drug payment rates to average sales price (ASP) minus 22.5% — a nearly 30% change from its previous price of ASP plus 6%. In December 2018, the U.S. District Court of Columbia held that the cuts were outside of CMS’s authority. However, there’s not yet a consensus on how the ruling will be carried out.
In early 2018, CMS reduced drug payment rates to average sales price (ASP) minus 22.5% — a nearly 30% change from its previous price of ASP plus 6%. In December 2018, the U.S. District Court of Columbia held that the cuts were outside of CMS’s authority. However, there’s not yet a consensus on how the ruling will be carried out.
The court did not motion to give immediate refunds to healthcare facilities that experienced the cuts, fearing that it may wreak financial havoc on the Medicare Hospital Outpatient Prospective Payment System (OPPS).
As of February 2019, no final decisions have been made in the court’s order. This leaves several questions unanswered, including:
- Will CMS appeal the decision?
- When will affected healthcare organizations be reimbursed for the cuts?
- Will CMS implement cuts that were scheduled to take place on January 1, 2019?
Controversy Continues
One of the key controversies surrounding the 340B program is accusations that hospitals simply aren’t using these savings to improve care for underserved populations. Instead, many large hospital systems are furthering their acquisitions and mergers initiatives, using the additional money to purchase independent physician practices.
Among the dissenters is the Large Urology Group Practice Association (LUGPA), which further argues that growing hospital systems are monopolizing the market. This limits patient choice and ends in patient charges increasing by as much as one-third for the same services.
Closing Thoughts
There is significant uncertainty for the future of 340B. Many experts expect that CMS will appeal the federal court’s 2018 decision, which means that this issue likely won’t see resolution in the near future.
We can expect to hear an update in mid-February, as the court ordered both parties in the ruling to submit a briefing on their recommended remedy. These briefings were submitted in late January, with each party having the opportunity to respond to each other within two weeks.
As the regulatory debate continues, so too do the voices of dissent as to whether 340B is truly benefitting U.S. patients and how issues may trickle down through pharmacy contracts.
In the time being, pharmacies can stay connected through memberships to industry organizations like AlliantRx.
If you’re not an AlliantRx member, learn more about joining us here.
